After completing a successful thesis focused on a novel pathway in cell division, at the Cancer Research UK Manchester Institute, Elvan Böke realized that she needed a career change. This decision to explore new lines of research brought her, after three years at Harvard Medical School in the USA, to the Centre for Genomic Regulation (CRG). Here, she set up and has been directing the Oocyte Biology and Cellular Dormancy group since 2017. It is a small group with two PhD students and a Master’s student, all women, but they are expanding. They will soon incorporate their first postdoctoral researchers.
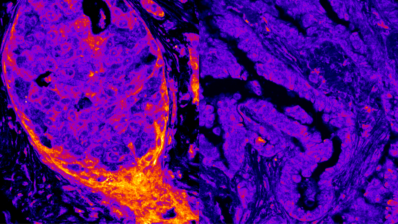
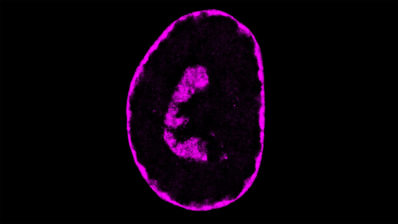
During her stay in Harvard, Elvan learned about the Balbiani Body, a discovery that changed her research life. This non-membrane-bound compartment is a key feature of dormant oocytes in many animals, from spiders to humans. It contains most of the organelles in dormant oocytes, and is stable only in these structures —it disperses once oocytes are activated.
“The Balbiani Body is a non-membrane-bound compartment that contains most of the organelles in dormant oocytes”
Böke immediately wondered how this solid structure could exist in the middle of the oocyte cytoplasm without a membrane. What was keeping everything stuck together? She demonstrated that, in frogs, this ‘glue’ is a protein called Xvelo, which forms an amyloid-like aggregate around organelles. “Amyloid-like aggregates are known for their key role in Alzheimer’s and ELA disease. We usually think about amyloid as pathological aggregates but there are also physiological amyloids”, she explains. These assemblies, once they have finished their mission, dissolve. But if there are problems and they do not dissolve, they can become pathological very quickly.
Oocyte particularity and the importance of the Balbiani Body
Oocytes are formed four months after conception. “When a female is born, all her oocytes are already inside her”, Böke explains. “Think of any other cell in our body: skin cells, fibroblasts, take your pick. Most of them don’t have a 40-year life span, like oocytes do. And if you look at those cells that do live for 40-50 years, such as neurones, they accumulate a lot of problems, including mutations and protein aggregations.” But oocytes cannot have these kinds of errors. If they are activated and start to develop a new human being, “this has to be from a fresh start; you cannot really have an old baby”, Böke points out. The Balbiani body could be the one that helps keep all the organelles and cytoplasm in the oocytes “young and healthy” during this extended period of time.
“When a female is born, all her oocytes are already inside her, so they have a 40-year life span or more, yet they cannot afford to accumulate mutations”
Working at the PRBB and clinical applications
Böke expresses her satisfaction at working in an organisation like the Biomedical Research Park of Barcelona (PRBB). “First, the core facilities are amazing, especially the animal facility. It’s really great, the staff provide everything we need. Also, as the PRBB is a multidisciplinary institution, it is easier to establish new collaborations with researchers with different backgrounds and points of view.”
Böke expects to stay at the CRG for nine years, the maximum time junior scientists can stay at the centre. She would be more than satisfied if her lab was able to identify the protein that holds the Balbiani Body together in mammals.
Such a discovery could have a direct impact on infertility treatment, one of the most common problems in the 20th century. “If we find the analogue of Xvelo in humans and we see, for example, that its levels decrease with age, we could explain fertility problems due to ageing. Nowadays women want to have kids at thirty five plus, and oocytes do eventually decline with age.”
The second clinical application may be in the treatment of Alzheimer’s. “If we understand how physiological amyloid-like aggregates dissolve we may be able to apply that knowledge to pathological amyloids”, concludes Böke.