Climate change is favoring the conditions for the reproduction and dissemination of infectious diseases typical of tropical climates, such as Zika, Chikungunya or Dengue, which are transmitted by mosquito bites. The viruses causing these diseases are of special interest because they can cross the blood-brain barrier (BBB) and penetrate the brain, where they can cause irreversible damage. In addition, if the Zika virus infects during pregnancy, it is also capable of penetrating the placental barrier (PB), causing damage to the baby’s brain, premature birth or even miscarriage.
The NOVIRUSES2BRAIN project, which has given the UPF €1.6 million from the European FETOPEN-Horizon 2020 program, aims to find a drug that is able to cross both the BBB and the PB in order to attack viruses that reach the brain or the fetus. It was launched almost four years ago and it involves researchers from the Department of Medicine and Life Sciences at Pompeu Fabra University (MELIS-UPF), the Institute of Molecular Medicine in Lisbon and Synovo GmbH in Tübingen.
We spoke with David Andreu and Toni Todorovski, researchers at MELIS-UPF, to learn more about the project.
Why is it important to find this kind of antiviral?
In the last 10 years, viral pandemics such as the SARS-CoV-2 , influenza, or the Zika epidemic that hit South America between 2015-2016, have been increasingly common. And, although research with antivirals is intense, since 1963 only 90 drugs have been approved for 9 human viruses. This is suprising when compared to the treatments that exist to treat diabetes or blood pressure, for example.
More alarmingly, the arsenal of treatments to fight viruses affecting the brain is even more limited. One of the reasons is that, although there are antivirals that work relatively well in vitro, they are unable to cross the BBB; something understandable, since that is what this barrier is for, to prevent anything from entering the brain. The problem is that some viruses make it… but antivirals do not, and this makes them ineffective.
So the main objective of the project is to get through the brain barrier?
Indeed. With this project we are not aiming to discover a new antiviral drug; those already exist. What we are looking for is to find a new way of administering antivirals so that they can reach the virus and attack it effectively. So the problem we have to solve is getting to a virus that has crossed these brain or placental barriers. Once we know how to get to it, we have drugs to eliminate it.
We could say that we design “smugglers” who, with a backpack full of existing antivirals, are able to cross through places where it is almost impossible to cross. So that, once on the other side, the package (the antivirus) deploys its potential and acts where it is needed.
“Our goal is to explore the viruses that can cross either the BBB or the BP and to propose solutions that direct the drugs to where these viruses are. Although this is an ambitious goal that no one has tackled before, our first results are promising”
To achieve the goal we have defined three phases of the project:
- Development of high-throughput vector + drug (smuggler + backpack) conjugates, the main area of work from MELIS-UPF.
- In vitro assays to test the penetration of the conjugate through both BBB and PB, and the efficacy of the antiviral component in these cases.
- In vivo assays of the conjugates with the highest antiviral potential in the previous phase. This is the phase with the greatest activity at present, and the most complex one, because eventhough there are good results in vitro, there are usually many surprises when moving to in vivo.
And how do you manage to get this new treatment through the barrier?
The treatment consists of a conjugate, i.e. the union of a drug with antiviral properties and a vector responsible for transporting it.
- The drugs, in our case, are porphyrins, broad-spectrum antivirals but unable to cross the BBB or the PB.
- The vehicle, or vector, are cell penetrating peptides (CPP), which are able to cross both the BBB and the PB effectively, but do not have antiviral properties on their own.
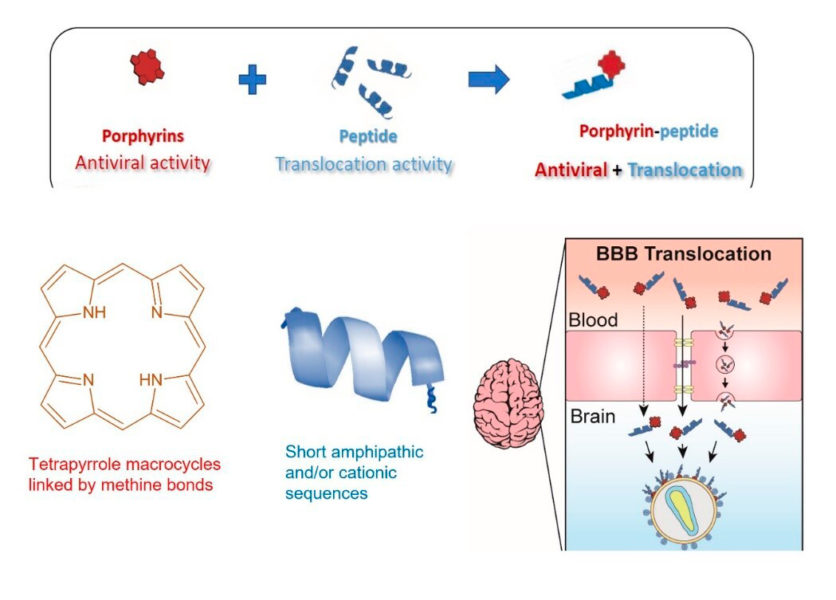
Our work has consisted of linking these two molecules to be able to deliver the drug in sufficient quantities to where the virus is found, something that seems simple, but which we have been working on for about three years…!
Why is it so difficult?
By combining 6 peptides, 5 types of porphyrins and a connector, we have designed and produced 24 different conjugates. It is a design in which we have taken into account the binding position of the porphyrin in the CPP structure, the distance between the two elements, etc. to achieve an optimal molecule with a good translocation rate and at the same time able to maintain and display antiviral properties. Another essential and relatively unpredictable aspect is toxicity, which can derail some candidates…
What results have you obtained and what are the next steps?
Our project is basic science: to design effective conjugates both in vitro and in an in vivo mouse model, which will allow us to propose new therapeutic strategies against viruses that affect both the brain and the fetus. In vivo studies currently underway indicate that we have a winning conjugate and a runner-up. Specifically, the results are very good against the Zika virus. But in addition, some conjugates are multivalent, with effect on other viruses such as HIV or SARS-CoV-2. Based on the results against Zika, we have already patented several of these new molecules designed in the laboratory and, from there, pharmaceutical companies may come into play to develop these molecules as drugs. Although we are still far from a multivalent antiviral.