A study led by researchers from the Hospital del Mar Medical Research Institute (IMIM) discovers a key piece in the treatment of some of the most aggressive cancers, which will allow them to be treated more efficiently. It is a protein that gives tumor cells resistance to treatments such as chemotherapy and radiotherapy, the IKKα kinase.
Until now it was known that this protein is expressed as a response to DNA damage. When there is a break in the double strand of DNA, the IKKα kinase is activated; it then activates another one, ATM kinase, which then starts phosphorylations of several key proteins that ultimately activate the “DNA damage checkpoint”. This checkpoint stops the cell cycle until the DNA has been repaired. Thus, IKKα kinase is responsible for stopping the cell cycle in order to repair DNA or, if necessary, to cause programmed cell death or apoptosis.
In this study, the authors have discovered the routes that regulate the expression of the IKKα protein and that trigger this whole process.
The role of oncogenes
When carrying a genetic mutation that increases their expression, oncogenes cause the transformation of normal cells into tumor cells. Researchers have discovered that an oncogene present in the most aggressive cancers, BRAF, is responsible for activating the IKKα kinase when the cell’s DNA is damaged – for example, when we tryto clinically combat metastatic tumors through chemotherapy or radiation therapy. This kinase then repairs the damage caused in the tumor cell’s DNA. This means that this protein would be “protecting” the tumor cells from the treatment to eliminate them, that is, it confers resistance to the treatment.
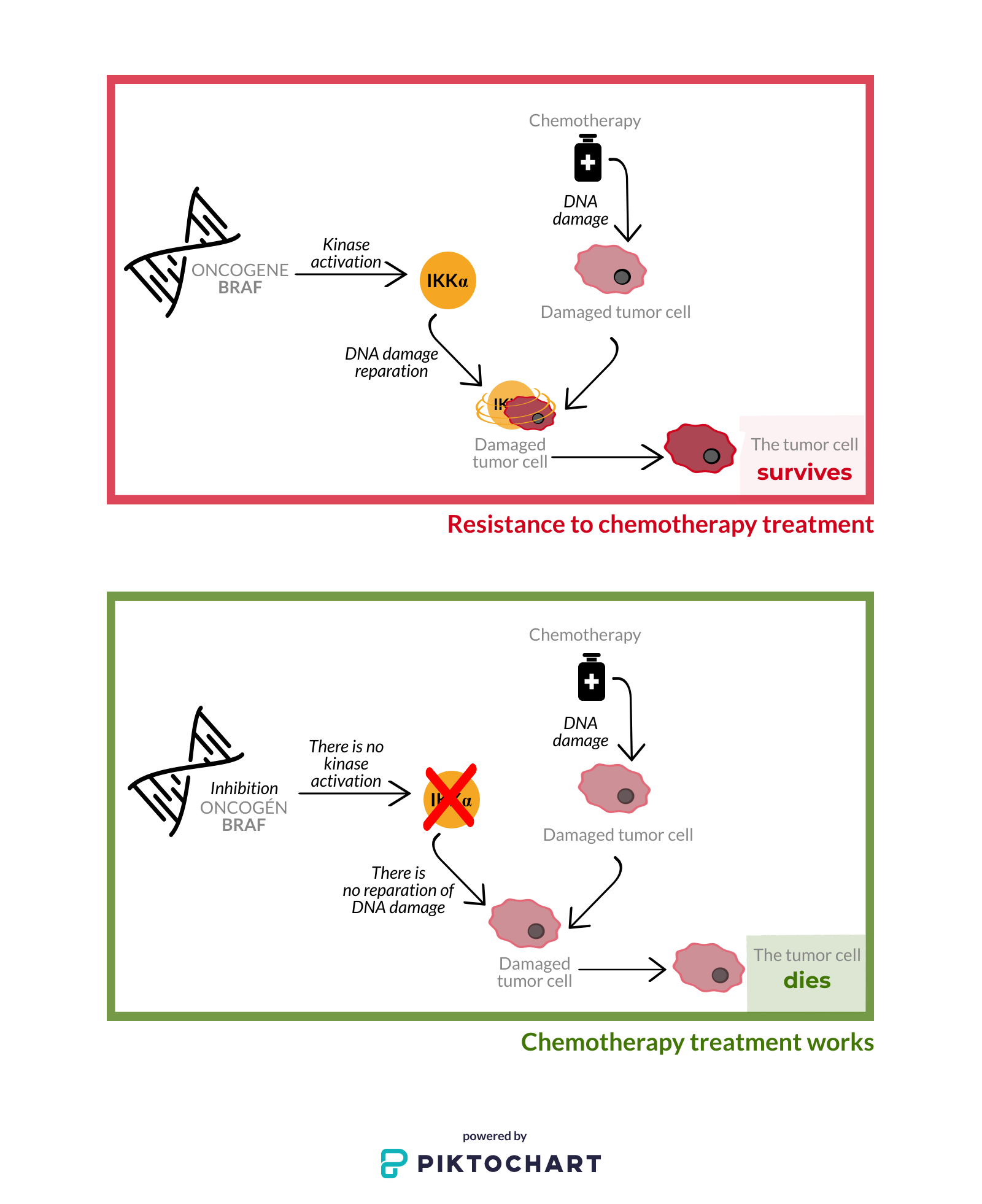
Knowing the role that this protein plays in the treatment of aggressive cancers, the researchers wondered whether its inhibition would “unprotect” the tumor cells against clinical treatment, facilitating their destruction. This has now been demonstrated in their study.
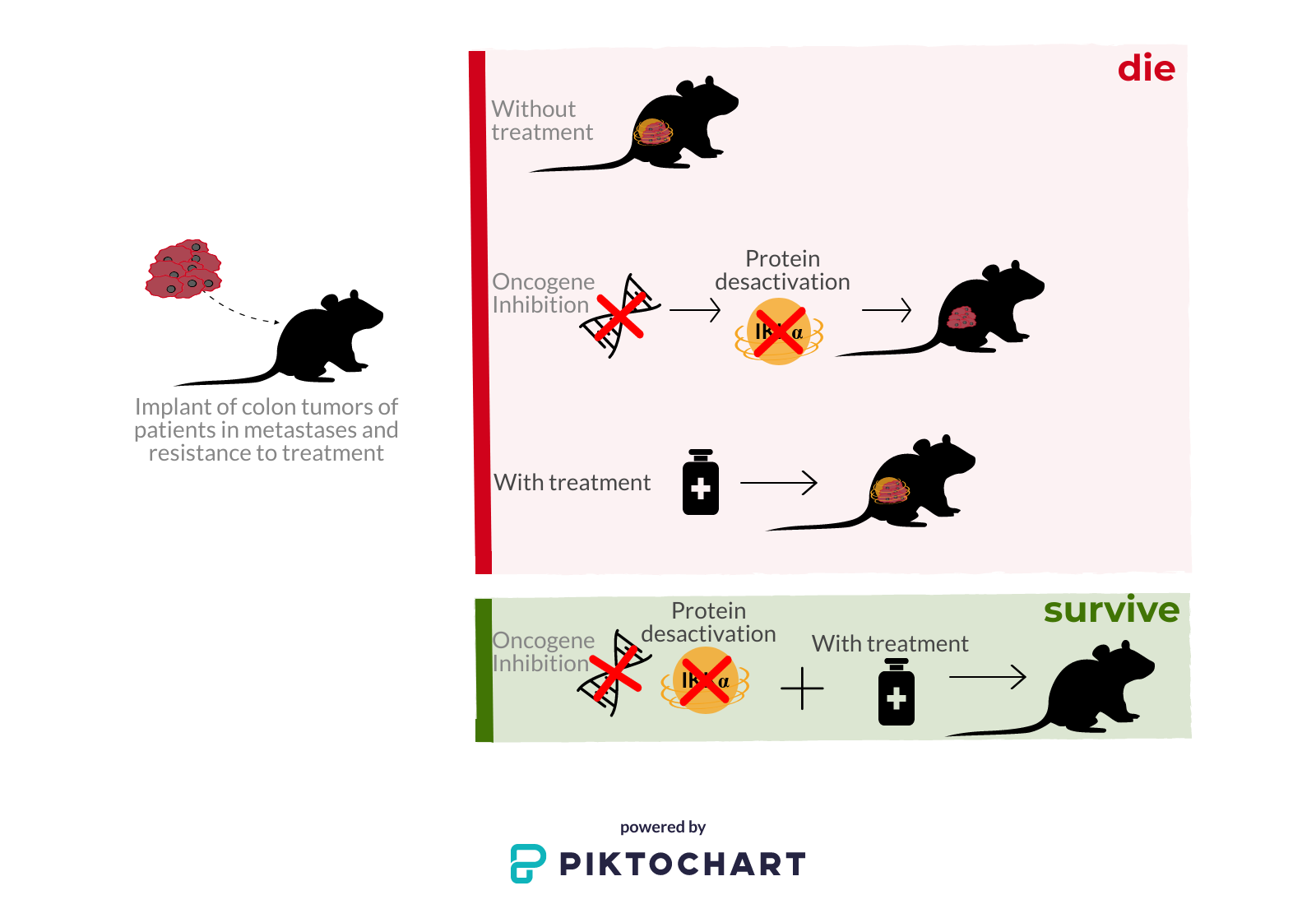
To do so, they implanted, in mice, colon tumors of patients with metastases and resistance to treatment, and subjected groups of mice to different conditions:
- No treatment
- With BRAF oncogene inhibitor (which activates the IKKα protein)
- With chemotherapy treatment
- With chemotherapeutic treatment and with BRAF oncogene inhibitor (which activates the IKKα protein)
The only surviving group was the last one, which received both chemotherapy and inhibition of the activity of the IKKα kinase by means of a drug that is currently used in clinical practice. Thus, the researchers, led by Lluís Espinosa, have been able to demonstrate that the combination of chemotherapy with the elimination of the activity of the IKKα protein, makes the tumor more sensitive to treatment and thus easier to combat.
Beyond mice
Discovering that the IKKα protein confers chemotherapy resistance to tumor cells, and knowing which oncogene is responsible for its activity, is a great advance in the cure of aggressive cancers. For this reason, the authors now intend to transfer this discovery to humans through a clinical trial with metastatic cancer patients in order to confirm the efficacy results that have already been observed in mice in this study, as well as to characterize possible side effects.
The study has been carried out by the IMIM Stem and Cancer Cell Group and doctors from the Medical Oncology and Pathological Anatomy services of the Hospital de Mar. Researchers from the Catalan Institute of Oncology, the Bellvitge Biomedical Research Institute (IDIBELL), the Center for Biomedical Research in Oncology Network (CIBERONC), members of the Proteomics Unit of the Centre for Genomic Regulation (CRG), and The Francis Crick Institute in London have also collaborated.
Hear Lluis Espinosa, leader of the study, explaiing what they did in this video.
Colomer et al., IKKa Kinase Regulates the DNA Damage Response and Drives Chemo-resistance in Cancer, Molecular Cell (2019), https://doi.org/10.1016/j.molcel.2019.05.036