A team led by Eva M Novoa at the Centre for Genomic Regulation (CRG) has discovered a mechanism that allows the heart to switch between two states: one of basic maintenance, and one of ‘fast loading’ in situations of high demand.
The lab has found that heart and muscle cells have specialised types of ribosomes that are different from those in the rest of the body.
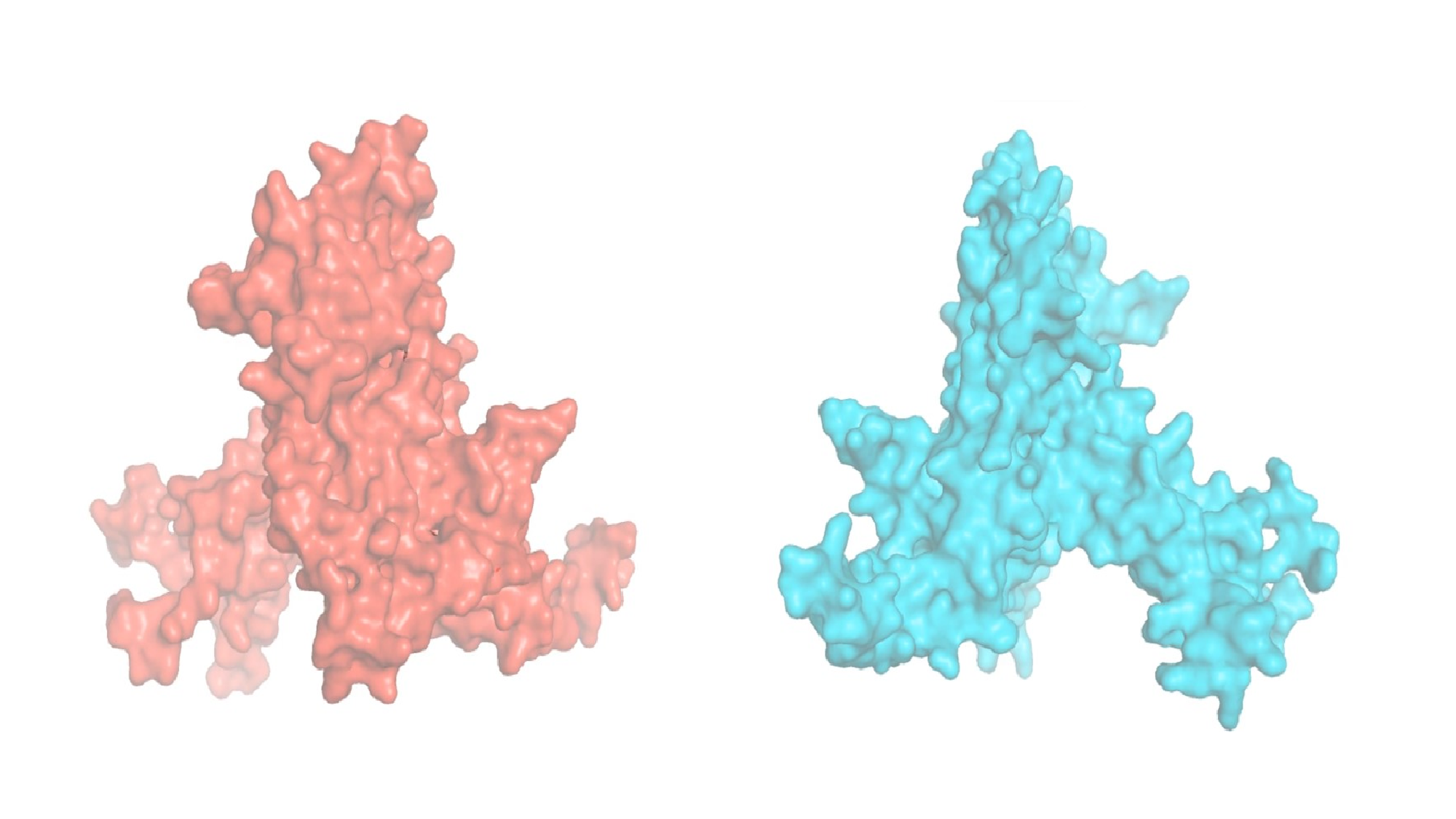
Ribosomes are the ‘protein factories’ of cells, and they are made of proteins themselves, including one called RPL3.
What they have discovered in this new study is that, in adult heart and muscle cells, ribosomes use a slightly different protein, RPL3L. The two variants of the protein only differ in the ‘tail’, the last part of the protein. But this difference makes the ribosome surface different so that it binds to other proteins and receptors.
Thus, the research group has also found that, when there is a cardiovascular disease such as a heart attack, or after heavy exercise, cardiomyocytes (the cells that make the heart contract) replace the RPL3L in their ribosomes with RPL3. This change means that the ribosomes can now physically contact the mitochondria – the cell’s energy factory – and increase energy levels. The change, which is reversible, occurs between 6h and 4 days after stroke or exercise.
The more energetic version of ribosomes (with RPL3) is also present in foetal heart and muscle tissue, researchers have found.
“When we are born, our heart needs a lot of energy to grow, but when the heart has fully matured, they switch to RPL3L, the basic maintenance state”
Ivan Milenkovic, CRG PhD student
Ivan Milenkovic, first author of the study and PhD student, explains why they believe that the adult heart and muscles do not, by default, have the most energetic version: “We think the cells may be adjusting mitochondrial energy-generating activity in resting conditions to decrease levels of free radicals, which are dangerous by-products of mitochondrial metabolism”.
The study, which started in the CRG lab led by Novoa, ended up as a collaboration with other labs in Spain, Germany, the Netherlands and Australia. And during the pandemic, when, explains Milenkovic, “I had to learn how to dissect ventricles through Zoom!”
Congratulations to the whole team!
Ivan Milenkovic et al. Dynamic interplay between RPL3- and RPL3L-containing ribosomes modulates mitochondrial activity in the mammalian heart. Nucleic Acids Research, gkad121, https://doi.org/10.1093/nar/gkad121
Published: 07 March 2023