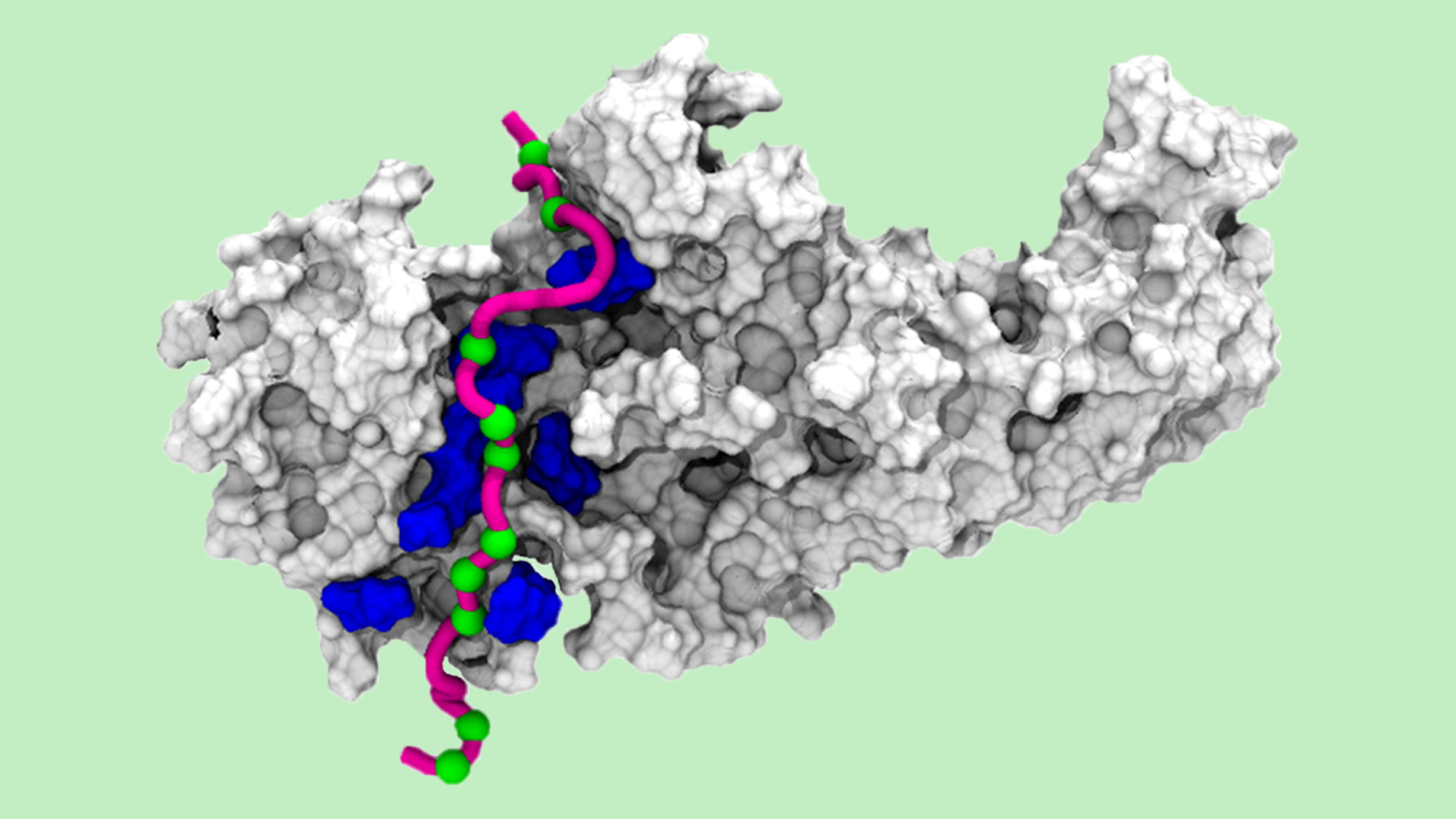
The GPCR Drug Discovery research group at the Hospital del Mar Medical Research Institute (IMIM), led by Jana Selent, has sent us this picture created by large-scale computer simulations, showing a 3D arrestin molecule ‘reading’ a specific phosphorylation pattern.
This is the result of a recent study done in collaboration with the Indian Institute of Technology in Kanpur. The researchers have deciphered the way β-arrestins change their three-dimensional structure in response to different phosphorylation patterns, which in turn modulates the main intracellular signalling cascade.
“We have discovered the underlying molecular mechanism of how arrestin reads and translates a specific phosphorylation pattern into a cellular response”
Jana Selent, head of the IMIM GPCR Drug Discovery group
The response of a cell to external stimuli is mediated by a type of membrane bound proteins called G protein coupled receptors (GPCRs). The signalling process triggered by the cell when stimulated is as follows:
- An extracellular stimulli (smell, taste, hormone, light, a drug…) arrives to the cell.
- This leads to the phosphorylation of the GPCRs tail. Depending on the stimulus, the phosphorylation pattern varies, altering the cell response.
- β-arrestins bind to the GPCRs tail and read the phosphorylation pattern. As the group has now deciphered, different phosphorylation codes induce distinct rotations in the arrestins’ 3D structures.
- According to their 3D structures, arrestins modulate intracellular kinases, which determine the cell’s behaviour by leading to gene expression, cell survival or cell death.
In the image, the phosphorylated sites of the ‘pink’ GPCR tail (seen as green spheres) are read by specific sensors in the arrestin molecule (blue areas in the picture). This information is then translated into a specific arrestin conformation which triggers further downstream signaling events.
“Our molecular findings create new opportunities to rethink current treatment approaches and to design novel therapeutic strategies for important diseases such as cancer or Alzheimer’s“, says Tomasz Stepniewski co-author of the study.
Would you like to see your photo here? Please send us pictures related to science or the PRBB to ellipse@prbb.org.
Dwivedi-Agnihotri et al. “Distinct phosphorylation sites in a prototypical GPCR differently orchestrate β-arrestin interaction, trafficking, and signaling“ Science Advances 11 Sep 2020 : EABB8368.