“The human genome devotes much more space to regulating genes than to the genes themselves”, says Jorge Ferrer, a researcher at the Center for Genomic Regulation (CRG). Indeed, only 1 to 2% of the genome are protein-coding sequences. Further knowledge of the areas of the genome that apparently have no function could provide us with more information on how diseases occur. A new CRG study, in collaboration with Imperial College London, has analyzed precisely one of these regions, identifying a new regulator of the activity of a gene that causes diabetes.
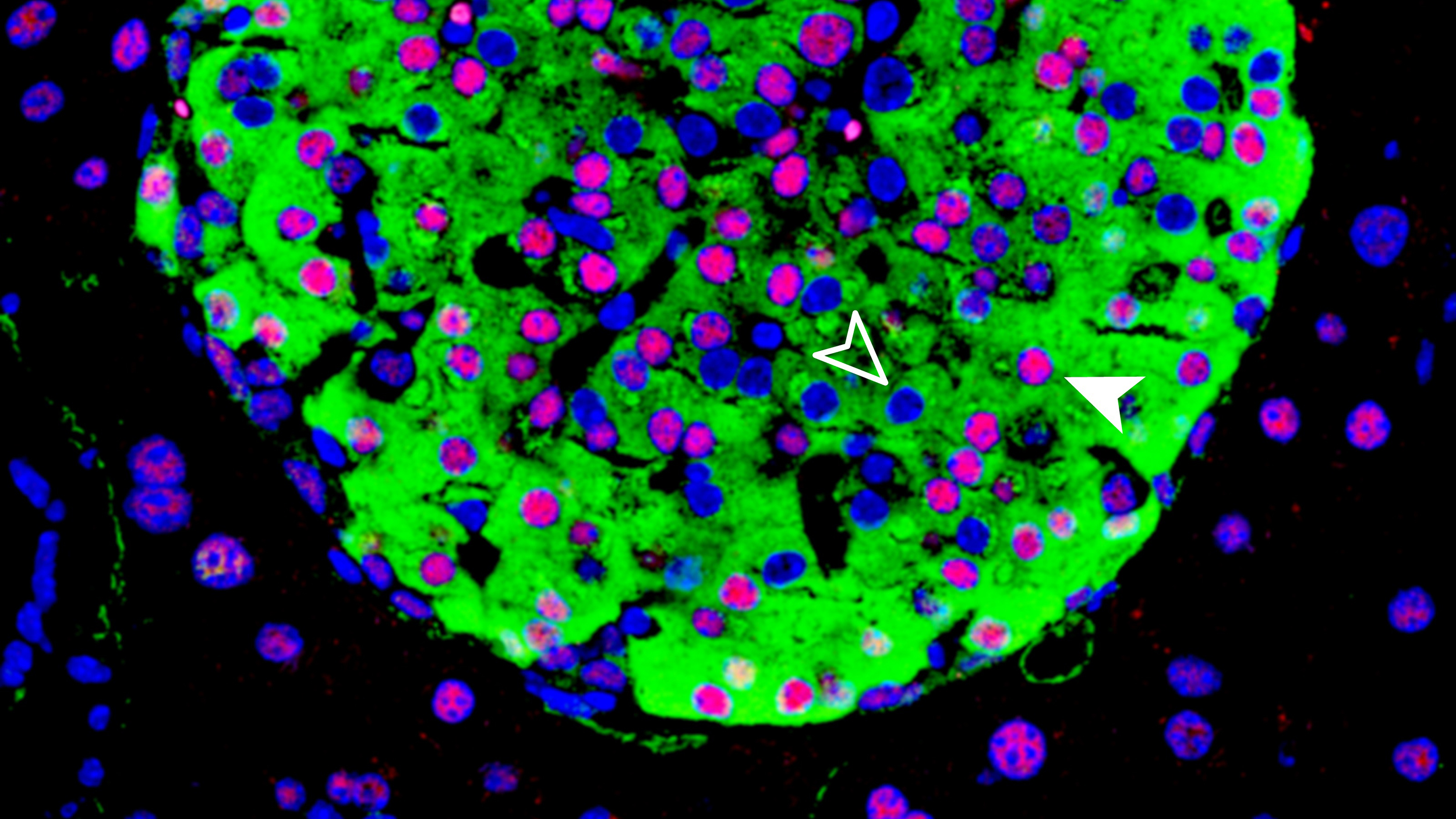
HNF1A is a gene expressed in many cells of the organism, but when expressed in the pancreas it produces hepatocyte nuclear factor 1 alpha. This protein plays an important role in the development of β-cells, which are responsible for producing the hormone insulin, the blood sugar levels regulator. A mutation in the HNF1A gene causes the patient to develop maturity onset diabetes of the young (MODY).
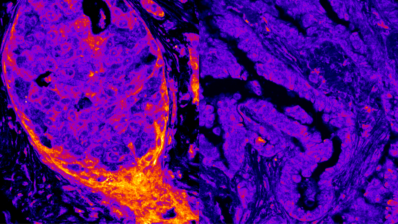
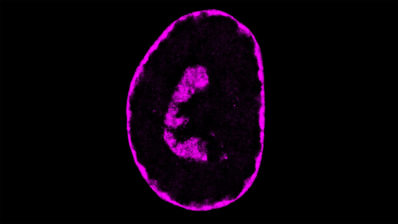
To better understand how HNF1A is regulated, the scientific team has analyzed the surrounding genome sequence in human and mouse models and identified the HASTER element. This sequence acts by moderating the intensity of production of an IncRNA, a long non-coding RNA molecule. When the HNF1A gene produces a lot of protein, the HASTER region controls it by reducing its activity. Conversely, when there is a reduction in the transcription of the gene, HASTER adjusts the intensity by increasing protein production.
“We have named HASTER as a stabilizer, in contrast to other DNA regulatory elements such as enhancers, promoters and silencers”
Jorge Ferrer, CRG
The study also shows that HASTER is a critical element, as a mutation in this region in mouse β-cells “has consequences comparable to deleting HNF1A”, according to Jorge Ferrer. There is still much to discover about these “meaningless” genomic sequences that could lead us to provide answers to how diseases work and open new paths towards new therapeutic targets. “There are tens of thousands of IncRNAs in the human genome, most of which have no known function. This is probably just the tip of the iceberg”, concludes Ferrer.
Beucher, A., Miguel-Escalada, I., Balboa, D. et al. The HASTER lncRNA promoter is a cis-acting transcriptional stabilizer of HNF1A. Nat Cell Biol 24, 1528–1540 (2022). doi: 10.1038/s41556-022-00996-8