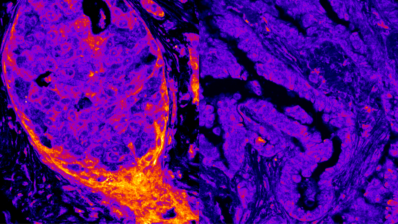
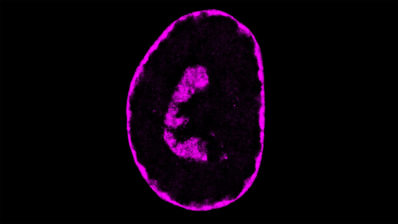
Similarly to other neurodegenerative diseases, protein aggregates are seen in the overwhelming majority of amyotrophic lateral sclerosis (ALS) patient brains and spinal cords. But, as is the case with the other diseases, it is not clear whether the aggregates are actually causing the disease.
Protein aggregation: cause or consequence?
In order to check that, a group of researchers at the Centre for Genomic Regulation (CRG) and IBEC have taken a thorough approach. Using a technique called deep mutagenesis, they took TDP-43, the protein that aggregates in the motor neurons of nearly all ALS patients, and created over 50,000 versions of it, each with a different mutation. They then introduced each of them in yeast cells and quantified their toxicity.
Surprising results
Surprisingly, the mutated proteins that caused aggregation, were the less toxic ones for the yeast, while the most toxic versions tended to be in liquid form. According to Ben Lehner, leader of the study, this is the exact opposite of what they would have expected based on the current understanding of these diseases. “It looks like aggregation actually protects the cells, rather than harms them” concedes Benedetta Bolognesi, co-first author of the article – together with André Faure – and currently a new junior group leader at the IBEC.
“Of course this has been done in yeast, which is a model system – we don’t know whether in humans there are other things at play… but this experiment does tell us that the aggregate itself, that solid thing, is not killing a eukaryotic cell. And that’s important, because every single paper on neurodegenerative diseases, such as Alzheimer’s or Parkinson’s, starts by saying “insoluble aggregates or plaques are the hallmark of disease”; which is true, but that doesn’t mean it’s the cause”.
“It’s possible that the harmful forms are intermediates of the aggregation process, but they may also be generated independently from the aggregates; they may not be related at all. We have not measured that”, caution the authors.
The authors created >50,000 mutants of TDP-43, the protein aggregated in amyotrophic lateral sclerosis (ALS) patient brains. They found that the aggregate itself does not kill a yeast cell.
The Italian researcher is now going to check whether the same thing (that liquid forms are harmful and aggregated forms are not) is true in mammalian cells. If it is, this might be a game changer for neurodegenerative diseases like ALS or Alzheimer’s, whose treatments have so far targeted the aggregation without success.
“What I like about this paper, and about the method used, is that because we look at all the possible mutations we can see some patterns which we would miss looking at one mutation at a time”, adds Bolognesi.
Genetic mutations can tell a protein’s structure
The method used, deep mutational scanning (DMS), involves creating thousands of mutant variants of the same protein and then analysing them – and it has indeed proved to offer new insights into protein structures and how they affect cells. Its use to work out the structures of proteins was recently published by the Lehner laboratory as a proof of concept, and the TDP-43 study has applied it.
Deciphering the 3D structure of proteins is usually done by one of several methods, such as X-ray crystallography, NMR spectroscopy or cryo- electron microscopy (cryoEM), but they all have limitations and tend to be expensive. Computational prediction from the amino acid sequence is not so costly, but it’s not always precise.
The researchers from this CRG lab took a cheaper and simpler approach: taking a protein, mutating every single amino acid in it, and then seeing how each mutation affects the growth of the cells where it is present. By finding pairs of mutations whose effects combine in unexpected ways, and assuming that these are likely to be physically close to each other in the folded protein, they can infer the protein’s 3D structure.
“Although DMS could be considered a mature experimental method, there is currently a great need for easy-to-use computational tools to analyse and interpret the resulting data as we did in this paper. This is why we are developing DiMSum: freely available software for processing DMS data”, adds André Faure, who led the data analysis in the TDP-43 paper.
Benedetta Bolognesi, Andre J. Faure, Mireia Seuma, Jörn M. Schmiedel, Gian Gaetano Tartaglia & Ben Lehner. The mutational landscape of a prion-like domain. Nature Communications volume 10, Article number: 4162 (2019)
Jörn M. Schmiedel & Ben Lehner. Determining protein structures using deep mutagenesis. Nature Genetics volume 51, pages1177–1186 (2019)